It seems that once you get to “a certain age” (around 50, give of take a few) you can expect stiffness and pain getting out of bed in the morning. After you move about for a while the pain subsides, but never completely disappears. Just sit at your computer for a couple of hours, and your lower back will remind you that something is amiss. Other joints don’t escape the ravages of age. In some people the knee joint becomes stiff and painful when walking or running. In others, the shoulder joints become painful.What went wrong? And what can be done about it?
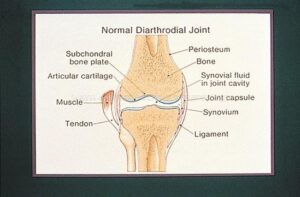
Normal knee joint. Source: The American Rheumatological Association, Image Bank
This picture shows a normal knee joint, but the basic anatomical features are the same in all joints. The space between the bones is separated by cartilage, a sponge-like rubbery tissue made up of interweaving fibers of collagen type II proteins richly decorated with sugar molecules called glycosamingoglycans (GAG). These molecules are important to understanding how cartilage works. Collagen type II is extra-strong, more than skin collagen (collagen type I). The sugar molecules attached to the core protein of GAG attract water, keeping the cartilage well-hydrated. The joint is bounded by a thin membrane, synovial membrane, that constantly secretes fluid, the synovial fluid, which fills the spaces between the fibers and also forms a thin layer of fluid on the upper and lower surfaces of the cartilage. So now we understand how cartilage functions: the rubbery tissue provides a cushion and the synovial fluid provides lubrication, to prevent damage to the cartilage by the moving surfaces of the bone.
When we age the synovial membrane loses its it structural integrity and the amount of fluid it secretes is reduced. This in turn reduces the lubrication between bone and cartilage, with the result of its progressive destruction, until in extreme cases we get bone-on-bone friction. This is the basis for osteoarthritis, the “wear and tear” disease of aging. Sometimes, inflammatory cells attack the synovial membrane and cause its destruction. The effect on the cartilage is predictable -it gets destroyed, just like in osteoarthritis. This is the basis for rheumatoid arthritis. Source: MedicineNet.com
Source: MedicineNet.com
Current treatments
Mild-moderate arthritic pain can be treated with simple exercise. One of the best exercises for low back pain is walking. I walk the dog firat thing in the morning, so both of us benefit ( or as cliché-prone folks like to say, win-win). I also do stretching exercises for the back, and this is enough for me to avoid taking any medications.
If this is not enough, the second line of defense is NSAID (pronounced EN-said), or non-steroidal anti-inflammatory drugs. These include aspirin, ibuprofen (such as Motrin, Advil), and naproxen (Naprocyn, Aleve). Should you take acetaminophen (Tylenol) for arthritic pain? Tylenol is pretty good for pain relief, but it lacks a crucial property: it is not anti-inflammatory. NSAID does both.
For severe cases there are new drugs that inhibit the inflammatory mediators (cytokines), but they are not cost-free. They can compromise the immune response and increase susceptibility to infection.
What about glucosamine/chondroitin sulfate supplements?
The only credible study (the GAIT study), was funded by NIH, not by industry, and included 1,583 patients. Here are the main results.
- Overall, there were no significant differences between the treatments tested and placebo.
- For a subset of participants with moderate-to-severe pain, glucosamine combined with chondroitin sulfate provided statistically significant pain relief compared with placebo—about 79 percent had a 20 percent or greater reduction in pain versus about 54 percent for placebo. According to the researchers, because of the small size of this subgroup these findings should be considered preliminary and need to be confirmed in further studies.
- For participants in the mild pain subset, glucosamine and chondroitin sulfate together or alone did not provide statistically significant pain relief.
Surprising? Not at all. Just consider: these compounds break down in the acidic environment of the stomach. They also get digested in the small intestine. Whatever escapes these hurdles gets digested in the blood. How much reaches the joints? Several studies failed to find any.
Surgery, anyone?
Not to slight the rheumatologists and orthopedic surgeons, there is a surgical procedure called microfracture, which consists of making tiny incisions in teh cartilage, with the hope that the repair mechanism would stimulate formation new cartilage tissue. good idea, except that it doesn’t work as advertised. The wound is repaired by fibroblasts, which secrete collagen type I, like in any scar tissue. This type is weaker that the cartilage-specific type II, which is secreted by chondrocytes, specially dedicated to cartilage formatiom.
and now for something completely different
Not very encouraging so far. At most we can slow down the destructive process with NSAID, but we cannot bring back our healthy cartilage. But there is glimmer of hope, I am happy ot say. A recent paper in PNAS by a group of bioengineers at Johns Hopkins headed by Jennifer H. Eliseef, describes what looks like regeneration of new cartilage is within reach.
In the laboratory, the researchers created a nanofiber-based network using a process called electrospinning, which entails shooting a polymer stream onto a charged platform, and added chondroitin sulfate to serve as a growth trigger. After characterizing the fibers, they made a number of different scaffolds from either spun polymer or spun polymer plus chondroitin. They then used goat bone marrow-derived stem cells (a widely used model) and seeded them in various scaffolds to see how stem cells responded to the material.
Elisseeff and her team watched the cells grow and found that compared to cells growing without scaffold, these cells developed into more voluminous, cartilage-like tissue.
The investigators then tested their system in an animal model. They implanted the nanofiber scaffolds into damaged cartilage in the knees of rats, and compared the results to damaged cartilage in knees left alone.
They found that the use of the nanofiber scaffolds improved tissue development and repair as measured by the production of collagen type II, a natural component of cartilage, and much more durable than collagen type I, which makes up scar tissue.
When can we use it?
Not next week or next year. Rats are not humans, and clinical trials can take several years. But when this, or another artificial tissue matrix does reach the market we’ll all be able to get up in the morning and go out for a run with no pain. Life could be as wonderful as it used to be.