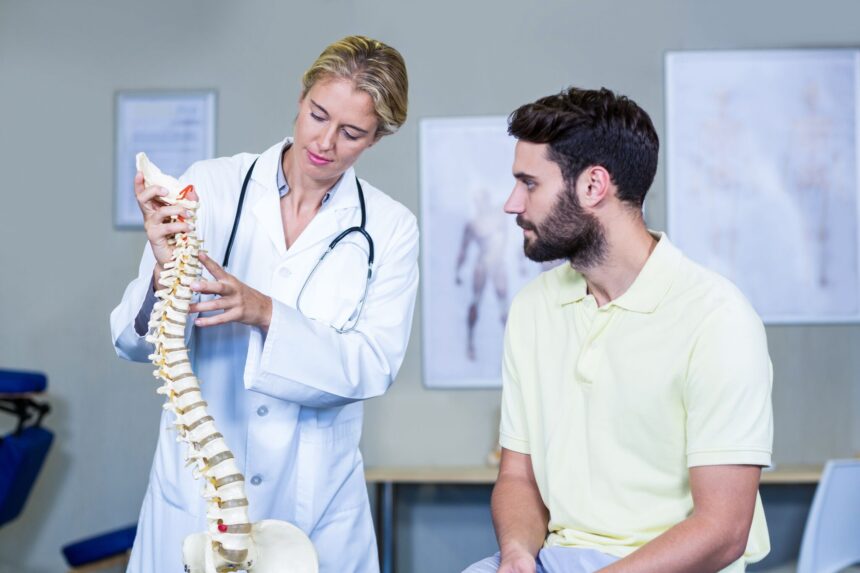
With the increasing prevalence of degenerative disc disease (DDD) and chronic back pain patients seeking minimally invasive medical procedures, the artificial disc replacement (ADR) market is expected to grow exponentially. Per research from the Global Market Insights, the artificial disc market is actually suspected to surpass $4.5 billion by the year 2024. Thanks to modern technology paired with new product development, the 3 million U.S. cases of degenerative disc disease and chronic low back pain now have an innovative option other than the once gold standard, spinal fusion. Improvement in artificial disc replacement (ADR), or total disc replacement (TDR), clinical trials created an increase in this surgical procedure among surgeons and patients alike. Why?
The Specifics
Because the purpose of artificial disc replacement surgery is to stabilize the patient’s spine while preserving the patient’s natural movement of range and motion, ADR provides an additional benefit as opposed to spinal fusion surgery. The artificial disc used for artificial disc replacement (ADR) allows the patient to move freely without pain or strain while supporting the spine’s natural range of motion with increased flexibility. The patient can also expect to resume normal activities faster because ADR’s recovery is traditionally shorter than those who undergo spinal fusion. This is because spinal fusions are invasive procedures (but you already knew that). That said, ADR has become the most preferred operation prospect for patients experiencing severe chronic back pain.
The Market
Lumbar (lower back) artificial disc replacement has beheld constant growth since 2004 and is expected to continue to increase. In fact, the artificial disc market received over $240 million in 2015 alone. At the same time, the cervical (neck) artificial disc replacement market share retained over 65 percent of total revenue and is suspected to exceed $3 billion by 2024. On top of it all, the metal implants used in ADR have seen significant gains, with expectations to exceed $4 billion by 2024. Research from Global Market Insights reveal, ?Lack of uniform and adequate reimbursement, high procedure cost, and expensive devices [may] hamper artificial disc market growth over the forecast period,? however, when comparing ADR to spinal fusion among patients with chronic low back pain, a recent study revealed that the results favored ADR at five years. With the growing need for cost-effective artificial discs, this should drive the market to grow further as it becomes a more mainstream treatment option.
The Location
In 2015, Germany seized over 20 percent of the artificial disc market share and is expected to grow by 15 percent. Demographic trends, advanced healthcare infrastructure, low cost of artificial disc replacement should feed industry growth. Over in China, with its advanced health care infrastructure with an increasing trend regarding medical tourism and cost-effective surgical operations, the artificial disc market will see a robust 24 percent compound annual growth rate (CAGR). The Japanese artificial disc market received more than 25 percent of their local revenue share. It’s anticipated to see a 20 percent CAGR because of large disposable income, modern health care infrastructure, as well as the technological advancement of ADR products. On the international level, Global Market Insights, Inc. says Healthcare Revenue Cycle Management Market size is supposed to exceed $100 billion by 2024. So, let’s uncover how surgical approaches, ADR, and spinal fusion, held up in patients with DDD over a five-year follow-up period. Will you agree with the market research that supports ADR while providing the patient with an increased quality of life and additional pain relief? Or, will you side with the once gold standard spinal fusion? Like we already said, let’s find out.
The Study
To evaluate the endurance, effectiveness, and safety of artificial disc replacement (ADR), also known as total disc replacement (TDR), a team of pain specialists including Dr. Jack Zigler, an orthopedic spine surgeon at Texas Back Institute in Plano, Texas, as well as Dr. Matthew Gornet an orthopedic spine surgeon at Orthopedic Spine Center of St. Louis and experts at Cornerstone Research conducted a meta-analysis of the available randomized controlled trials with published five-year outcomes comparing ADR to spinal fusion. 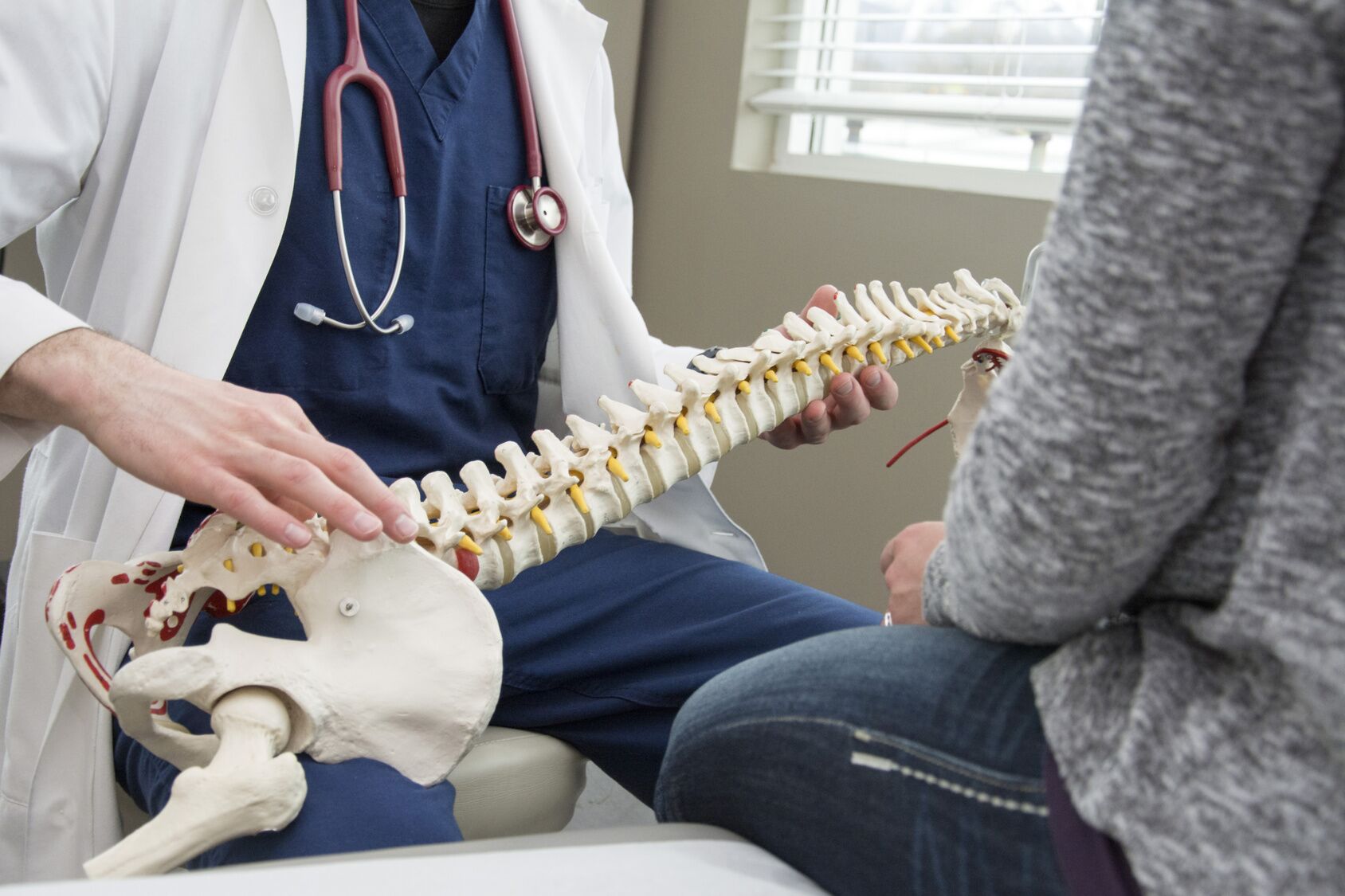 The patients were those diagnosed with chronic lower back pain from single-level lumbar Degenerative Disc Disease (DDD). This is what you need to know.
The patients were those diagnosed with chronic lower back pain from single-level lumbar Degenerative Disc Disease (DDD). This is what you need to know.
The Method
The authors examined all of the trial results between patients who underwent ADR surgery with those patients who had a spinal fusion in single-level lumbar DDD as well as their five-year outcomes using data from PubMed and Cochrane Central Register of Controlled Trial’s medical directories. Using well-respected statistical methods, the authors pooled together the evidence from all four studies to make broader, higher impact conclusions about how ADR stacks up to fusion in the long-term. Based on the Discussion section of the manuscript, the authors were not surprised at the strength of the results in favor of ADR. The outcomes involved included these measurements:
- The Oswestry Disability Index (ODI). A tool physicians use to estimate a patient’s permanent functional disability. Patients answer questions in 10 sections, which are each scored separately (0 to 5 points each) and then totaled ? the maximum score being 50. The lower the number, the less pain the patient experiences.
- Back pain scores. Applied to determine the mean change in the patient’s back pain score from before surgery all the way until the five-year follow-up period.
- Patient satisfaction. Scored to test the patient’s willingness to choose the same surgery again.
- Reoperations. Examined and defined as device-related failures resulting in the subsequent surgical interventions of a reoperation, revision, removal, or supplemental fixation. Was a second surgery needed?
- All surgeons who perform ADR are required to report product problems, if they learn any of their devices may have caused or contributed to a death or serious injury. This is known as Device-related serious adverse events (SAEs), which was also examined during the five-year follow-up period.
All investigations from the four analyses were executed using a random-effects model, also called ?mixed models? or ?variance components models.? These models allow for a patient?s individual characteristics to be considered rather than pooling their results into an average sum with other participants. This removes any biases while allowing the result to be a real variation that notates biological and environmental factors rather than just an analytical trick to test the variability of patients? surgical responses. Analyses were reported as relative risk (RR) ratios and mean differences (MDs). Sensitivity investigations were administered for different outcome definitions, high loss to follow-up, and high heterogeneity (diverseness).
The Bias Factor
All four studies showed the same risk for bias. As per sound statistical methods, the authors completed sensitivity analyses to control any potential bias throughout all four studies.  All of the studies were randomized, which means that the surgeon investigators who implanted the devices did not know if the patient would be receiving fusion or ADR until after the patient was selected.
All of the studies were randomized, which means that the surgeon investigators who implanted the devices did not know if the patient would be receiving fusion or ADR until after the patient was selected.
The Outcome
The meta-analysis incorporated four randomized controlled trials reporting on the five-year outcomes of ADR versus fusion testing the five outcomes above. The results included: ADR was shown to be statistically better than fusion ? ODI success ADR patients were more likely to have a clinically relevant improvement in their ODI scores than fusion patients at five years (p = 0.05) ADR was shown to be numerically but not statistically better than fusion ? Back pain scores There was no affiliated improvement in any patients? back pain scores whether the DDD patient received lumbar ADR or a spinal fusion during the five-year follow-up (p = 0.25) ADR was shown to be statistically better than fusion ? Patient satisfaction Patients reported greater satisfaction with an implanted ADR device rather than spinal fusion during the five-year follow-up (p = 0.01). ADR was shown to be statistically better than fusion ? Reoperations Patients who received ADR had a lower risk of reoperation (p = 0.002). The need for reoperations resulting from device-related malfunctions was consistent in all four studies. Treatment of lumbar DDD with ADR produced a 48 percent decrease in the chance of reoperations as compared to treatment with spinal fusion.
The Conclusion
Based on the study’s evidence, degenerative disc disease patients who received lumbar disc replacement saw more long-term strides in pain relief including motion preservation making ADR a beneficial alternative treatment to spinal fusion. “ADR is an effective alternative to fusion for lumbar DDD. It offers several clinical advantages over the longer term that can benefit the patient and reduce health care burden, without additional safety consequences,? the authors concluded.
Summing Up
- When contrasted with traditional therapeutic approaches such as physical therapy, exercise, activity limitation, and meditational pain management, ADR exhibited equal if not higher positive results at five years.
- Opting for ADR decreases the patient’s chances of any adjacent discs degenerating further increasing the patient’s chances of reoperation without pain relief.
- Patients in the meta-analysis who underwent ADR were notably more likely to be satisfied with their treatment option at five years.
- Improving healthcare expenses, favorable reimbursement policy structures, growing disposable income and higher success rates of artificial disc replacements should spur further business growth.