Dental sector professionals have a wide range of impression materials at their disposal and can choose the product best suited for each specific treatment. Due to recent technical developments in the field of dentistry, the latest impression materials guarantee more accurate final results and less discomfort for patients. One of the most important recent advances in dentistry is in the field of impression materials. In this article we examine the characteristics needed and recommended techniques for use.
Alginates for dental impressions
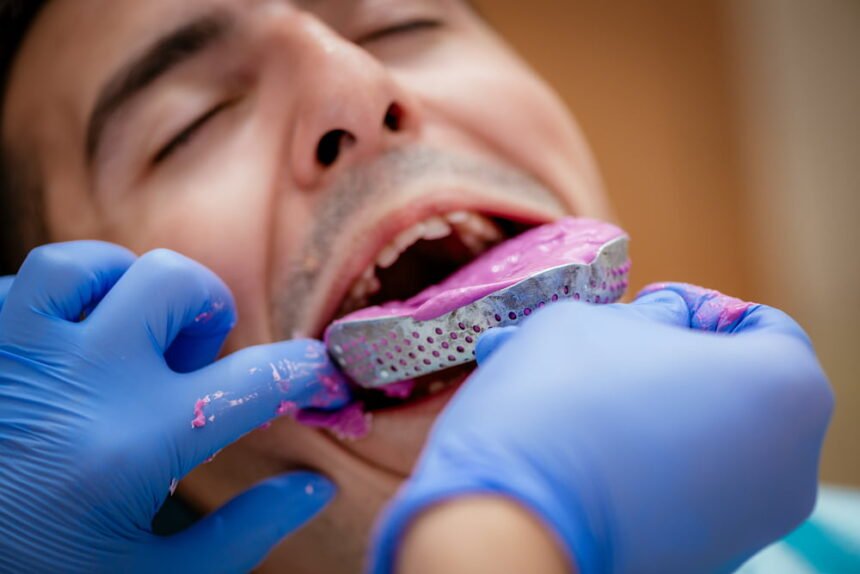
Alginate impression materials are indispensable for many common procedures. They require collaboration between the dental practice and the laboratory. Since they can be easily mixed and are well tolerated by patients, these materials are widely used to take preliminary and primary impressions and in the production of study models.
To ensure correct use of these compounds, however, it is important to follow certain procedures. Read our useful hints on how to take a dental impression and achieve high-performance results.
Correct dosing of water and powder is one of the most important aspects to bear in mind. The water-to-powder ratio determines the properties of the material and influences manufacturer’s specifications like times and compound stability. If the dentist chooses to mix manually, he should measure out the powder first and add the water afterwards. Powder should also be shaken prior to use to distribute the mix more evenly. Many dentists prefer automatic to manual mixing. This recent development in dental technology reduces the number of bubbles in the mix and therefore reduces macroscopic defects in the impression and the stone model.
Once the mix has been prepared, the dentist can choose from various types of tray to suit the clinical case and their own personal preferences. Two common ways to improve retention between the alginate and the impression tray are to use a perforated tray or to apply an adhesive compatible with the chemical and physical properties of the alginate.
If a stone model cannot be cast immediately, alginate impressions can be preserved by eliminating excess water, placing them in a sealed polythene bag, and storing them at ambient temperature in a container in the dental practice. Take care not to leave impressions standing in disinfectant solution too long, as extended soaking can damage alginate materials. Hydrocolloids are hydrophilic by nature and expand when immersed in disinfectant or water.
These impressions are some of the most important tools dentists can use. It is important to use them correctly.
Addition and condensation silicones
Silicones are among the most reliable and accurate modern impression materials. Addition silicones like Zhermack’s Hydrorise System range are extremely accurate and reliable while condensation silicones like Zhermack’s Zetaplus System range guarantee excellent final hardness and reduced risk of tearing during removal from the mouth.
When dosing addition silicones, measuring spoons should never be mixed up. Never measure catalyst with the base spoon or vice versa. Contamination between the two components can prevent correct curing of the final compound. Likewise, latex gloves should not be worn when working with addition silicones. Latex interferes with the platinum catalyst and inhibits polymerisation. Nitrile or vinyl gloves should be worn instead.
The dosing phase is of fundamental importance with condensation silicones too. An incorrect amount of catalyst can impact the chemical and physical properties of the material and lead to poorer performance. Using more catalyst than specified in the instructions, for example, will reduce dimensional stability of the compound and shorten working time and time in mouth. Unlike addition silicones, condensation types can be worked with latex as well as vinyl and nitrile gloves without causing any alteration in their properties.
Do not use different materials for the impression tray and the preparation. If a condensation silicone is used as impression tray material and an addition silicone for the preparation, the surfaces may not adhere well, since the two materials have very different chemical structures.