Genomics X Prize will require teams to accurately sequence the genomes of centenarians.
Genomics X Prize will require teams to accurately sequence the genomes of centenarians.
 Genomics X Prize will require teams to accurately sequence the genomes of centenarians.
Genomics X Prize will require teams to accurately sequence the genomes of centenarians.
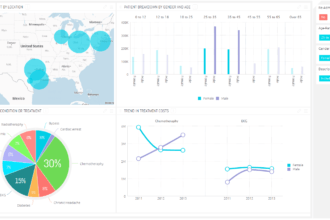
The X PRIZE Foundation revealed a number of major changes to its Archon Genomics X Prize, the $10 million competition designed to drive breakthroughs in the future of predictive and personalized medicine to make significant advances in human genome sequencing. Medco Health Solutions, Inc. (NYSE: MHS) has joined as the competition’s Presenting Sponsor. The X PRIZE Foundation and Medco stated the competing teams will be required to sequence 100 genomes of centenarians.
“The goal of this competition is to push the industry to develop, more accurate, faster and more cost effective sequencing technologies,” said Dr. Craig Venter, PhD, Co-Chair of the competition. “While many new technologies have been developed over the last decade there is still no technology that can produce a highly accurate, reproducible human genome usable for diagnostics and medical treatment. For genomics to truly impact health and diagnostic decisions for all of us, we need these technologies. We believe this competition will be the impetus to truly usher in the era of personalized medicine.”
According to the terms of the competition, a $10 million prize purse will be given to the first team that accurately sequences the whole genome of 100 subjects within 30 days for $1,000 or less per genome, at an error rate no greater than one per million base pairs.
To make the competition scientifically valuable and more meaningful to the general public, every team will be given the DNA of 100 centenarians to sequence. These 100 genomic pioneers, who represent a unique population of individuals who have survived their first 100 years of life, will be hereafter known as the ‘Medco 100 Over 100’ and will have donated their genome to this competition in order to further medical science.
DxTerity and Caliper collaborate
DxTerity Diagnostics, Inc. and Caliper Life Sciences, Inc. (NASDAQ: CALP) say they have entered into a co-development and co-marketing strategic collaboration that will allow researchers to perform DxTerity’s NEAT multiplex diagnostic assays on Caliper’s LabChip Dx instrument platform minimizing sample processing time without compromising data quality. Caliper’s future LabChip Dx instruments will include specific diagnostic identification, scoring and reporting software, developed in collaboration with DxTerity. The NEAT assays allow analysis of gene expression and detection of single nucleotide polymorphisms directly from blood or formalin-fixed paraffin embedded tissue samples. DxTerity’s lead clinical product in development is a blood screening test that measures a patient’s radiation exposure following a nuclear event, such as the recent disaster in Fukushima, Japan. The initial release of this product is scheduled for June 2012.
IN BRIEF…
New FISH technology test
Abbott (NYSE: ABT) announced that it has received FDA 510(k) for a new in vitro diagnostic test to aid in determining the prognosis of patients with acute myeloid leukemia (AML). Abbott’s Vysis EGR1 FISH Probe Kit detects a chromosomal deletion in bone marrow that is usually associated with an unfavorable prognosis for AML patients.
Quintiles acquires Advion
Quintiles said that it has signed a definitive agreement to acquire Advion BioServices, a bioanalytical laboratory providing Good Laboratory Practice (GLP) pharmacokinetic/pharmacodynamic testing and other services. The amount that Quintiles will pay was not released. Advion BioServices has more than 180 employees in three U.S. locations: Ithaca, N.Y., Manassas, Va. and Indianapolis.
Curetis adds to its series A round
German diagnostics company Curetis AG reports that it has raised an additional 9.6 million euros ($13.3 million) from investors to fund human tests of a system to identify infectious diseases. Forbion Capital Partners, a Dutch venture capital firm, led the expansion of the Series A financing round, which now totals 34.1 million euros. –Peter Winter