At $500,000 for the machine it is not cheap by all means and the price in the last  year just went up. The machine makes much of spine surgery minim
year just went up. The machine makes much of spine surgery minim
At $500,000 for the machine it is not cheap by all means and the price in the last  year just went up. The machine makes much of spine surgery minimally invasive.
year just went up. The machine makes much of spine surgery minimally invasive.
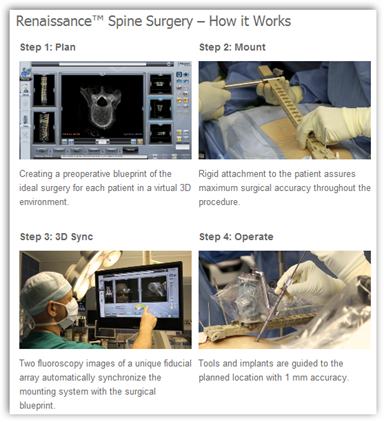
The video shows how the procedure works and and Mazor is from Israel. The robot is attached to the patient body and identified the most accurate position and surgical instruments can be inserted through the robot as well. Accuracy with the robot is much higher for placement. We all know that back surgery is one of the most risky types of surgery.
Mazor Robotics Ltd. (TASE:MZOR) has obtained US Food and Drug Administration (FDA) and EU CE Mark certification for its Renaissance product, the next-generation version of its robotic spinal surgical device, SpineAssist. 
The launch of the Renaissance is part of Mazor’s marketing strategy. The company has sharply raised the price of the SpineAssist in the past year, positioning it as a premium product that not only offers a quick return on investment, but also brings in new patients to hospitals with the device. Intuitive Surgical Inc. (Nasdaq: ISRG), which markets its da Vinci surgical system, a robotically assisted minimally invasive surgery device, has employed a similar marketing strategy.
Mazor receives FDA, CE approval for new spinal surgery robot – Globes