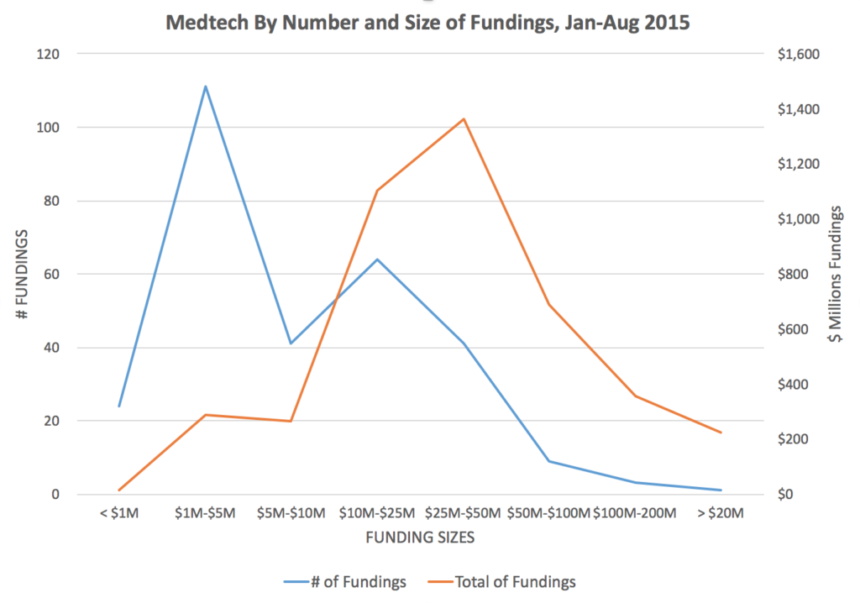
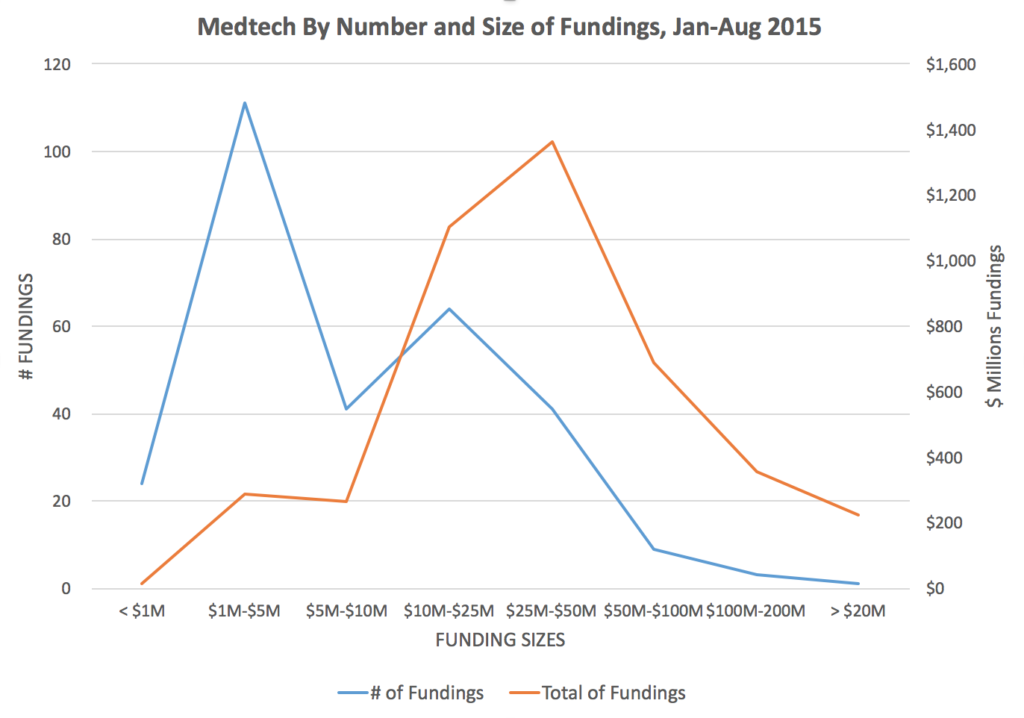
Fundings in medtech in 2015 follow a common pattern in that most fundings are between $1 million and $5 million — in 2015 thus far, we have identified 111 separate fundings in the $1M-$5M range — with the aggregate value of all fundings in this range being $286 million. However, the size category with the highest cumulative fundings is fundings at $25 million to $50 million, of which there were 41 separate fundings, reflecting an aggregate of $1.36 billion.
Fundings in medtech in 2015 follow a common pattern in that most fundings are between $1 million and $5 million — in 2015 thus far, we have identified 111 separate fundings in the $1M-$5M range — with the aggregate value of all fundings in this range being $286 million. However, the size category with the highest cumulative fundings is fundings at $25 million to $50 million, of which there were 41 separate fundings, reflecting an aggregate of $1.36 billion.
Source: MedMarket Diligence, LLC; see specifics in August 2015 fundings and historical fundings.