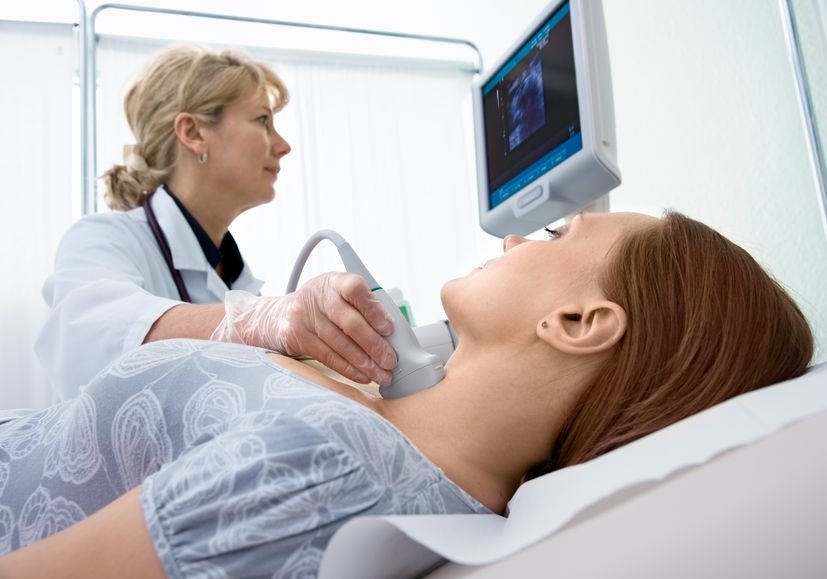
Roughly 57,000 people are diagnosed with thyroid cancer every year in the U.S. alone. Those who are able to catch the disease in its early stages have extremely high chances of making a full recovery when they have the thyroid surgically removed. However, there are nearly 100,000 people annually who actually end up having their thyroid removed when it wasn?t really necessary to do so. For those individuals, the surgery, as well as its associated costs and emotional effects, could have been avoided. That?s why experts have been working hard to create advancements in the realm of thyroid care, and at this year?s American Thyroid Association?s Annual Meeting, a new diagnostic tool was introduced that has the potential to eliminate unnecessary thyroid removals.
A Bit of Background on ThyroSeq V3
For the last 10 years, ThyroSeq has been combining the biggest scientific discoveries and advanced technology in an effort to help medical professionals give the most accurate diagnoses of malignant and benign nodules in the thyroid. One of the biggest advancements in thyroid care recently came with the study on ThyroSeq V3. ThyroSeq is a partnership between CBLPath and UPMC, and the results of this study were presented at the 87th Annual Meeting of the American Thyroid Association. The study was a multi-center, double-blind international study that was able to validate the latest version of ThyroSeq and its ability to more accurately diagnose cancer when there are nodules on the thyroid. The goal of ThyroSeq is to reduce the number of unnecessary diagnostic thyroid surgeries and to improve the ability of medical professionals to diagnose patients. And the study proves its success. ?Every indication is that we?ll be able to avoid countless unnecessary thyroid surgeries and offer more effective, individualized management of patients with thyroid nodules and cancer, based on this utting-edge genomic science,” said Yuri Nikiforov, M.D., Ph.D, Professor of Pathology at University of Pittsburgh Medical Center, and principal investigator of the multi-center ThyroSeq V3 trial.
Doctors on Board for Changes
Recently, thyroid experts have voiced their concerns regarding the many thyroid surgeries that are performed every year. They feel that one of the ways to start shifting the methods used to diagnose thyroid cancer would be to remove the term ?carcinoma? from one of the more common types of thyroid cancer because it actually isn?t invasive to other tissues and shouldn?t be considered cancer anymore. The hope is that, once the word cancer is removed from the name, surgery and radio iodine treatments that are ultimately unnecessary could be avoided. But whether or not the name is changed, better diagnostic procedures that make use of advanced technology are needed in order to reduce the number of unnecessary thyroid surgeries in the U.S.
Excitement Over ThyroSeq V3
After ThyroSeq?s study results were presented, experts became excited at the idea of being able to use ThyroSeq V3 in standard clinical settings as a trusted diagnostic method. Doctors now have a great tool that can be used across demographics and on benign and malignant nodules. That?s because V3 features a larger genomic classifier, which exhibits greater sensitivity and specificity when it comes to thyroid cancer detection. Overall, ThyroSeq V3 offers a procedure that could help to reduce both exploratory and diagnostic surgeries by as much as 60% amongst individuals who have thyroid nodules. It won?t be long before it becomes the standard of care in the treatment of these thyroid problems.