It sometimes feels strange to me that I am so interested in amyotrophic lateral sclerosis (ALS). I have no personal connection to this fatal motor neuron disorder. The only association I have had with ALS is I once worked in the area because of my job. Although it was a short period of time, it was a rewarding experience.
It sometimes feels strange to me that I am so interested in amyotrophic lateral sclerosis (ALS). I have no personal connection to this fatal motor neuron disorder. The only association I have had with ALS is I once worked in the area because of my job. Although it was a short period of time, it was a rewarding experience. Seeing the level of devastation of this horrible disease, I have never felt more grateful to be able to enjoy some of the most basic things in my life I used to take for granted—running after work, walking in the park, chatting with my friends, using a fork to eat and breathing the air. When ALS progresses, these basic things become difficult and eventually impossible for ALS patients to conduct. But the gratefulness is not the only thing coming out from my experience–I had the opportunity to witness how a group of patients, researchers and organizations collaborated with each other to innovate and advocate. The collective efforts, which have not yet led to finding an effective treatment, offer valuable insights for the pharmaceutical industry and healthcare system into how we should change our approach to deal with deadly conditions like ALS. When “Big Data,” personalized medicine and open science begin to reshape the future of medicine, the experiments taking place inside the ALS community deserve our great respect and everyone in the industry should take serious notes.

Stephen Hawking is also an ALS patient who has a very rare type of ALS that progresses much lower (Photo: hawking.org.uk)
ALS, also known as Lou Gehrig’s disease, is a progressive, debilitating, and fatal neurodegenerative disease. It causes dysfunction and death of upper and lower motor neurons in the brain and spinal cord. ALS is generally fatal within a median of 2-4 years of symptom onset.
In the last few decades, a lot of medical breakthroughs have turned many acute and severe diseases into manageable chronic conditions. But ALS remains a fatal disorder and there has not been any effective treatment to reverse the course of the disease. The only approved medicine is riluzole (Rilutek), which may increase survival by only 3 to 5 months.
Today, very few diseases like ALS speak about the fundamental meaning of “unmet need”—the need is so urgent that without it, people are dying, quickly.
While the industry has been searching for treatments for ALS, the ALS patient community has also been activated to participate in the process.
James Heywood is an important name for the ALS community. He is a co-founder of PatientsLikeMe, an online patient platform where people can connect with others with the same condition. The creation of the platform was inspired by James’ brother Stephen, who was diagnosed with ALS in 1998. The site was launched in 2004 and its first online community for ALS patients debuted in 2006, the year Stephen passed away. Unlike other online patient forums, PaitentsLikeMe was created not only to serve as a communication channel but also incorporate visions to provide solutions to some of the major challenges faced by ALS drug development.
The current ALS R&D status —no effective diagnostic tools, no identified biomarkers, continuous failures in drug development—emphasizes how ineffective the traditional drug development model is. With only a few years to live for most ALS patients, the traditional clinical trial model is not robust to support drug development for the deadly disease.
PatientsLikeMe, however, creatively uses crowdsourcing to measure patient outcomes in real time–a new way to look at clinical data. Registered users now can report their real-world experiences on more than 1,500 diseases. Through their profiles, they can keep a close eye on all vital information related to the disease management, such as monitor their health between doctor or hospital visits, document the severity of their symptoms, identify triggers, note how they are responding to new treatments, and track side effects. Additionally, they learn from the aggregated data of others with the same disease and see how they are really doing.
Although a lot of studies will be needed before we can say this new approach does bring value from a health care delivery perspective, with more than 100,000 registered users and being the largest online ALS patient community, PatientsLikeMe has already shown us the power of crowdsourcing and its potential to change the way we perceive drug development, clinical trials and patient communication. And some preliminary data have shown the value of patient engagement in clinical studies. In 2010, data gathered from the site showed the ineffectiveness of lithium, an investigational treatment for ALS. In 2012, James Heywood wrote another paper discussing data gathered around PatientsLikeMe users in the clinical trial that investigated dexpramipexole, a highly anticipated compound for ALS (unfortunately, the drug failed).
The platform has since attracted attention from the industry.
In February, the platform launched a program with Aetna, to help Aetna members improve the way they live with various health conditions. In the same month, it formed another partnership with the pharmaceutical company Boehringer Ingelheim to enhance its online patient community for people with idiopathic pulmonary fibrosis, a rare lung condition with no known cause, treatment or cure. These new initiatives highlights
These initiatives highlights how digital innovations originated from platform built for ALS patients can potentially increase productivity in healthcare delivery.
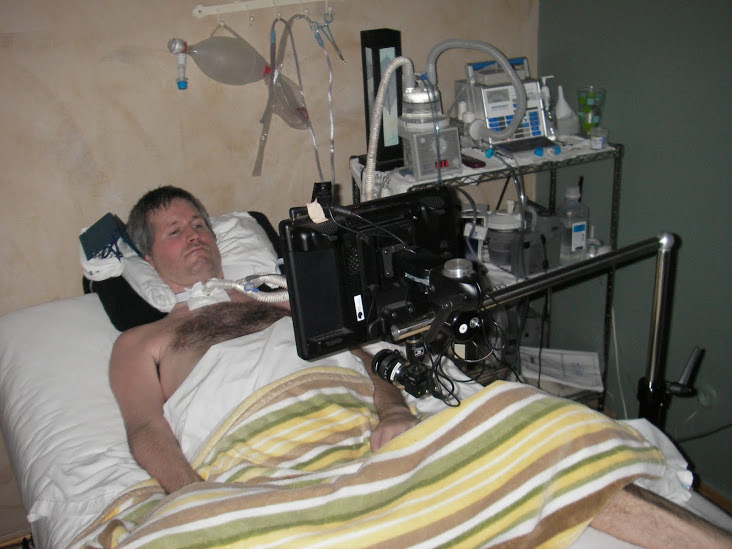
Eric was diagnosed with ALS when he was 36 (Photo:Friends4Eric.org)
Eric Valors is another name that struck me. He was diagnosed with ALS in 2004 when he just turned 36. By 2008, he has required 24/7 ventilation monitoring and care. But he has never stopped fighting. He was convinced, after research, sodium chlorite, a chemical used by water treatment plants is the active ingredient of NP001, an investigational drug for ALS. He injected the chemical (with help) and tracked how well he was doing on the drug. The DYI approach, although controversial, highlights challenges associated with traditional clinical trial design—is it moral to assign dying patients to a placebo-controlled group when a drug can potentially work? Eric’s story, which has been widely reported by media also reminds us with access to a significant amount of data , we don’t have enough knowledge to process, analyze and interpret them. The good news is that the Phase 2 results of NP001 showed possible benefit in ALS patients.
In the US, there are 30,000 people with ALS, and 7,500 new patients are diagnosed each year. The number may be small, but it is a reminder that we still don’t have any meaningful treatment for ALS. An active patient blogger will stop writing because the the author can no longer write. A charity dedicated to ALS will lose its spokesperson because he or she can no longer speak about the inspiring story about fighting the disease. Without effective treatment, every one of the newly diagnosis will die within just a few years. That is a cruel fact. Too cruel for us to accept in the second decade of the 21 century, when we are convinced technology can almost do anything.
After so many failed trials, researchers, regulators and pharmaceutical companies need to realize we need a brand new strategy to deal with ALZ. And they have the wisdom, support and understanding from all ALS patients.
May is ALS Awareness Month.If you’d like to know more about ALS and how you can help, below is a list of resources:
- ALS Association: http://www.alsa.org/
- Patients Like Me: http://www.patientslikeme.com/
- ALS Therapy Development Institute: http://www.als.net/
- Friends for Eric: http://www.friends4eric.org/home
- A Life Story Foundation: http://www.alifestoryfoundation.org
- Team Gleason: http://www.teamgleason.org/