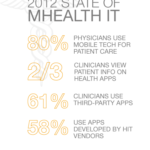
Like everyone else, I believe that we will eventually see smartphones playing significant roles in healthcare, but there are some hurdles that we need to clear.
Like everyone else, I believe that we will eventually see smartphones playing significant roles in healthcare, but there are some hurdles that we need to clear.
Scott Mace does a good job of summing them up in his article, “mHealth Trials Are Happening, Without the Clinic,” on Health Leaders Media. He wrote the article during the HIMSS’ mHealth Summit in Washington. Like he says, it’s easy to be dazzled by all the health apps that vendors are now offering. Medicine, as an industry, has always exercised caution before giving its blessings to drugs and treatments, and similar caution is in order before the widespread use of medical apps.
Mace says the elephant in the room that no one is talking about is government regulation because many of the apps provide disease diagnosis and treatment without a doctor’s involvement. So the question is (or perhaps should be) Does an app that diagnoses a medical problem practice medicine without a license? Hmmm. As Mace suggests, we probably won’t know until an app maker gets sued after someone suffers serious bodily harm or dies.
Speaking of regulation, California Representative Mike Honda has introduced a bill in Congress that would create an FDA Office of Wireless Health Technology. Rather than be concerned about the use of medical apps, Honda fears that FDA oversight could stifle innovation. He thinks an office focused on mobile medical technologies would bring these apps and smartphone devices to market more quickly. According to Mina Kim, whose article, “Bill Would Help FDA Assess Mobile Health Technology” on the KQED Web site, the proposed bill would also establish a program to help mobile app developers build their devices in line with federal privacy regulations. I hazard to say that creating this bureaucratic office will exceed the congressman’s estimate that operating costs would be a million dollars a year. No government office ever comes in UNDER projections. And, does the government need to be in the software business?
Meanwhile, Mace says the medical app industry doesn’t have much time left to show that it can police itself, or the government, perhaps this proposed FDA office, will impose even more requirements. What if given new regulations, consumers who are technology geeks decide to take matters into their own hands? People have been known to modify their smartphones – in fact, that’s how a lot of these iPhone devices and apps came about in the first place.